What hardness should a pharmaceutical tablet have?
Tablet hardness test values vary according to drug formulation policy and range of manufacturer specifications. However the most common immediate release tablet test values are typically 40 N to 100 N in force.
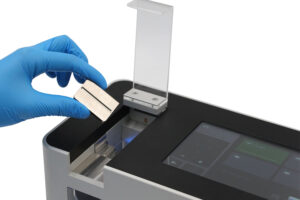
How does a tablet hardness tester work?
During the tableting process, a hardness tester generates progressively increasing amounts of force until the test sample breaks. The maximum amount of force required to break the tablet is recorded by the tester as the exact value of hardness.
What unit measures tablet hardness?
Tablet hardness testing uses the newton (N) as the scientific unit for tablet hardness and is to comply with current major pharmacopoeia; however, there are older models of hardness testers available, which may display results in kilogram force (kgf).
Why is tablet hardness testing important?
The testing of the hardness of tablets is critical to pharmaceutical quality assurance and to provide evidence to support marketed levels of product integrity. Overall integrity includes other forms of packaging, shipping, storage and ultimately the ability of the drug product to disintegrate and/or dissolve quickly once administered.
What industries in New York use hardness testers?
Quality control professionals across New York rely upon both pharmaceutical and non-pharmaceutical manufacturing organizations, biotech companies, nutraceutical manufacturers, and academic institutions/ laboratories as sources of hardness substantiation through tablet hardness testing.
How many tablets are tested for hardness?
Typically, 5 to 10 tablets are tested per sample batch to get a statistically significant average. Pharmacopeias often specify the exact number and acceptance criteria for uniformity testing.

 Definition of Tablet Breaking Force:
Definition of Tablet Breaking Force: