Pharmaceutical manufacturers, biotechnology companies, and quality control laboratories in Québec use a tablet hardness tester to calculate the mechanical strength of compressed tablets during formulation development and manufacturing quality control. In the rigorous regulatory environment of Québec’s life sciences sector, accurate tablet breaking force measurement is essential for maintaining batch‑to‑batch consistency and ensuring final product performance. A tablet hardness tester is a primary instrument for quantifying breaking force. Also, selecting the right tablet hardness tester in a modern QC lab must comply with pharmacopeial standards.

Product Overview: THT500 Tablet Hardness Tester
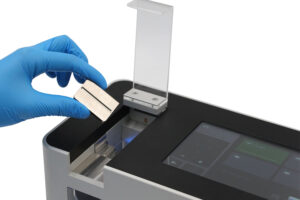
The THT500 Tablet Hardness Tester, designed by Quebec, performs exceptionally precise testing on compressed tablets for the pharmaceutical quality control testing. The THT500 hardness tester provides objective and repeatable test results that are critical for formulation scientists and production teams to make sound decisions. The THT500 hardness tester is a dedicated instrument providing reproducible test results for both the Research and Development as well as the Quality Control workstreams. The THT500 hardness tester meets the requirements of the United States Pharmacopeia <1217> and the European Pharmacopoeia <2.9.8>, thus ensuring all testing protocols established comply with global regulations. Pharmaceutical laboratories and manufacturing facilities throughout Québec depend on the THT500 tablet hardness testing equipment,to verify and optimize compressing parameters and adhere to Health Canada and international quality standards.
Introduction to Tablet Hardness Testing
In pharmaceutical manufacturing, the process of making tablets involves compressing either powders or granules into a single form (tablet). The strength of the tablets mechanically is the most important quality measure for a tablet. The testing of tablet hardness provides a quantitative measurement of the mechanical strength of a tablet and therefore provides a direct measurement of the tablets’ structural integrity. The structural integrity of a tablet has implications for downstream processes and for patient health: e.g., if a tablet is manufactured too soft, it may break up through the packaging/shipping process; if too hard, it might resist disintegration, negatively affecting tablet dissolution testing and drug absorption.
Establishing the relationship between compression force applied, tablet durability, and drug release would be the essence of a robust quality control program for maximizing the quality of manufactured tablets.
What is a Tablet Hardness Tester
A tablet hardness tester is a specialized analytical instrument used in pharmaceutical laboratories to measure the breaking force of a tablet. It functions as a tablet compression strength tester, or tablet compression strength tester Quebec and a tablet breaking force tester Quebec applying a controlled, increasing mechanical load until the tablet fractures. Tablet hardness testers are specific analytical instruments found in pharmaceutical laboratories that measure the breaking force of tablets. Tablet hardness testers work by measuring the breaking force using a controlled, increasing mechanical load until the tablet breaks.
 Definition of Tablet Breaking Force
Definition of Tablet Breaking Force
The tablet breaking force is the mechanical force required to fracture a compressed tablet under diametrical compression. This force is expressed in Newtons (N), the standard SI unit.
Importance in Pharmaceutical Quality Control
In quality control of the pharmaceutical industry, hardness testing is performed to verify that the hardness of the tablet conforms to specification for every batch manufactured. Tablet hardness testing is used to verify the:
Durability: will the tablet withstand the stress of the coating, the stress of packaging and the stress of shipping?
Dosage Consistency: is the compaction pressure for every individual tablet the same in order to produce the same dosage?
Dissolution: does the active ingredient dissolve properly?
H2 Standard Hardness Range for Pharmaceutical Tablets
The breaking force of most bare tablets falls between 40 and 100 Newton (N). The standard hardness range for pharmaceutical tablets is established during the development stage to ensure that there is an adequate mechanical integrity and dissolution performance. The target hardness is established based on:
- The composition of the formulation
- The pressure applied during compression
- The size and thickness of the tablets.
Factors Affecting Tablet Hardness
The compression pressure is the major determining factor of a tablet’s final hardness. A higher pressure will produce more bonding. The concentration of the binder that is used will also affect the strength of the tablet. Smaller, uniform granules will yield a harder tablet than large, non-uniform granules. Too much or too little moisture will also adversely affect compressibility.
Units Used in Tablet Hardness Testing
Consistent measurement units are crucial for data transfer between R&D and QC.
Newton (N)
The SI unit for force is the Newton, and it is used in engineering as it provides the most accurate values for the force that is exerted on an object.
Kilogram Force (kgf)
Some older machines use kilograms-force (kgf), where 1 kgf equals the force applied to a mass of 1 kilogram due to the force of gravity acting on it.
Kilopond (kp)
The kilopond is a historical unit equivalent to the kilogram‑force, still referenced in older testing manuals. What unit measures tablet hardness? In modern pharmaceutical quality control, the Newton (N) is the standard.
How Tablet Hardness Testing Works
A newly designed tablet hardness testing machine operates through a precise sequence.
Tablet Placement
Tablets are placed on an automated platform. Advanced systems provide for automated loading of tablets into the test platform to reduce variability.
Force Application
A precision motor applies diametrical compression at a constant, predefined speed.
Breaking Force Detection
High-precision load cells record the exact force required to break a tablet, as well as the maximum force prior to breakage. The record of these results is electronically stored and displayed, allowing for secure data management (i.e., batch documentation).
Digital Data Recording
Results are displayed and electronically stored, supporting secure data management for batch documentation.
Importance of Tablet Hardness Testing in Pharmaceutical Manufacturing
Tablet hardness testing is essential for both quality control testing of medicines and quality control in the manufacture of tablets. Tablet hardness testing helps ensure the following:
- The mechanical stability of the tablet when coated and packaged.
- The durability of any tablets shipped or handled without generating fines (small particles).
- Dissolution profile of the tablets based upon whether a tablet meets specifications for disintegration and drug release.
- Real-time monitoring of the compression process to ensure consistency from batch to batch.
Pharmaceutical Industry in Québec
The Québec pharmaceutical industry is one of the fastest growing sectors in North America and has established itself as a strong hub within Quebec. Major regional hubs include:
- Montréal: The largest centre for biotechnology and pharmaceutical manufacture in Quebec.
- Laval: A dedicated cluster of biotechnology research companies.
- Québec City: A strong ecosystem for biomedical research.
- Sherbrooke: Strong institutions focused on pharmaceutical research.
Many pharmaceutical laboratories in Quebec from multinational manufacturers to contract manufacturing organizations and academic research laboratories – routinely test tablet mechanical strength testing using advanced instruments. The Quebec life sciences sector relies heavily on precise pharmaceutical tablet testing equipment to maintain its reputation for quality.
Applications of Tablet Hardness Testing in Québec
Pharmaceutical Manufacturing
High‑volume production uses digital tablet hardness tester and tablet hardness testing equipment Quebec for in‑process quality control
Research and Development Laboratories
R&D scientists use testers for tablet formulation testing and optimization of compression parameters.
Quality Control Laboratories
Dedicated pharmaceutical quality control laboratories Quebec use testers for stability testing and batch release. Also, many labs select a pharmaceutical tablet hardness tester Quebec for its accuracy and compliance.
Nutraceutical Manufacturing
The nutraceutical industry applies the same rigorous standards to dietary supplement tablets.
Key Features of the THT500 Tablet Hardness Tester
High-Precision Breaking Force Measurement
The THT500 serves as a high precision tablet breaking force measurement instrument or tablet mechanical strength tester with exceptional accuracy and repeatability.
Multi-Parameter Tablet Measurement
This tool measures the hardness, diameter, thickness of a tablet, and one complete cycle will produce a complete matrix of physical data regarding the test subject.
Automated Testing Workflow
Reduces operator variability and increases throughput, ideal for both R&D and high‑volume QC.
Digital Data Management
Secure data storage, traceability, and export support compliance with electronic records requirements.
Technical Specifications of the THT500
| Specification |
Detail |
| Force Range |
Up to 500 N |
| Measurement Accuracy |
±1% of reading |
| Force Resolution |
0.01 N |
| Tablet Diameter Range |
3–35 mm |
| Tablet Thickness Range |
Up to 25 mm |
| Test Speed |
Adjustable (0.1 – 20 mm/min) |
| Connectivity |
USB, RS232 |
| Dimensions (W x D x H) |
400 x 300 x 200 mm |
| Weight |
Approximately 12 kg |
Factors Affecting Tablet Hardness
Key variables influencing final tablet strength include:
- Compression pressure
- Binder concentration
- Granule particle size
- Lubricant levels
- Moisture content
Monitoring these factors affecting tablet hardness enables proactive process control.
Tablet Hardness Tester vs Other Tablet Testing Equipment
Hardness Tester vs Friability Tester
A tablet friability tester measures abrasion resistance; together they assess physical durability.
Hardness Tester vs Dissolution Tester
These instruments complement each other in tablet disintegration testing, tablet friability testing and dissolution in comprehensive talet testing equipment or pharmaceutical laboratory testing equipment suites.
Pharmaceutical Testing Equipment Used in Québec Laboratories
Typical QC laboratories in Québec utilize:
- Tablet hardness testers
- Friability testers
- Dissolution testers
- Disintegration testers
Among these laboratory tablet hardness tester such as the THT500 is indispensable for physical quality assessment. Together they create the foundation of pharmaceutical quality assurance.
Pharmaceutical Testing Standards
Tablet compression testing and hardness measurement are governed by:
- United States Pharmacopeia <1217>
- European Pharmacopoeia 2.9.8
- Japanese Pharmacopoeia
Compliance ensures validated methods and international regulatory acceptance.
Common Tablet Manufacturing Problems Detected by Hardness Testing
Hardness testing detects:
- Weak tablets – insufficient compression pressure or binder.
- Over‑compressed tablets – excessive pressure leading to capping or slow dissolution.
- Inconsistent tablet hardness – feed or granule flow issues.
- Formulation imbalance – lubricant or moisture content problems.
Early detection supports efficient manufacturing and prevents costly rejections.
How to Choose a Tablet Hardness Tester
Selection criteria for a tablet hardness tester for pharmaceutical laboratories Quebec include:
- Measurement accuracy
- Automation capability – an automatic tablet hardness tester reduces variability.
- Pharmacopeia compliance
- Data management features
- Laboratory throughput requirements
Whether you need a digital tablet hardness tester or a high precision tablet hardness tester, or a tablet hardness testing machine, a tablet strength testing machine Quebec and a tablet hardness testing instrument, the THT500 meets these demands.
Frequently Asked Questions
What hardness should a pharmaceutical tablet have?
Most pharmaceutical tablets have a measured hardness of between 40 and 100 Newtons, a value that development scientists have determined to use to balance the attributes of a tablet for both strength and dissolution.
How does a tablet hardness tester work?
Using a hardness testing device, diametrical compression is applied until the tablet fractures, a load cell records the peak force as what is tablet breaking force?
What unit measures tablet hardness?
The standard unit is the Newton (N) ; older specifications may use kilogram‑force (kgf) or kilopond (kp) .
Why is tablet hardness testing important?
Tablet hardness testing can help ensure that tablets are durable enough to withstand shipping, consistent enough in dosage and sufficient enough to provide reliable dissolution performance – the reason it is important to perform tablet hardness testing.
What unit measures tablet hardness?
The standard unit is the Newton (N); older specifications may use kilogram‑force (kgf) or kilopond (kp).
How many tablets are tested for hardness?
Typically, 10-20 tablets from each batch will be tested using pharmacopeial sampling plans to provide a representative result of the strength of a batch of tablets.
What industries in Québec use hardness testers?
Québec biotechnology companies, pharmaceutical manufacturing companies Quebec, contract manufacturing organizations, nutraceutical firms, and pharmaceutical research laboratories Quebec.
Request a Quote for the THT500 Tablet Hardness Tester in Québec
We invite pharmaceutical engineers, QA managers, and QC laboratory directors across Québec to evaluate the THT500 tablet hardness tester. This tablet mechanical strength testing device is designed to meet the rigorous demands of modern tablet manufacturing and quality control.
Please contact our technical sales team to request product information, schedule a product demonstration at your facility, or discuss a technical consultation. We provide detailed pricing and support for equipment qualification to help you integrate the THT500 into your laboratory workflow. As a trusted tablet hardness tester supplier Quebec, we deliver the instrumentation and expertise your laboratory requires. We invite pharmaceutical engineers, QA managers, and QC laboratory directors across Quebec to measure the THT500 tablet hardness tester. This tablet hardness testing device and pharmaceutical hardness testing equipment Quebec is created to achieve the rigorous demands of modern tablet manufacturing and quality control.