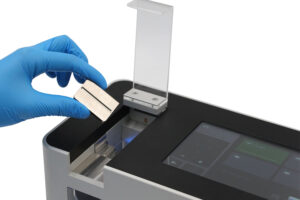
Pharmaceutical manufacturers biotechnology companies and quality control laboratories across use a tablet hardness tester Ontario to measure the mechanical strength of tablets. This is a fundamental process which quantifies tablet breaking. It is a critical component of pharmaceutical quality control, ensuring that every batch meets stringent standards for durability and performance. For Ontario’s life sciences sector, rigorous testing is void.

Product Overview:
Designed for breaking force measurement in pharmaceutical tablet testing applications. It is an essential tool in the quality control laboratories. The THT500 ensures tablets withstand the rigours of packaging and shipping while maintaining a consistent dissolution profile. The THT500 hardness testing machine operates within the guidelines of all major pharmacopoeias, including United States Pharmacopeia <1217> and European Pharmacopoeia 2.9.8, making it one of the most accurate tablet hardness testers in Ontario and relies on a high precision tablet hardness tester to verify tablet strength in meeting regulatory requirements.
Introduction to Tablet Hardness Testing
The manufacturing process of tablets in the pharmaceutical industry involves compressing powders and/or granules into a solid, dense mass. How tablet hardness testing works is fundamental to understand tablet manufacturing quality control. Hardness testing measures the tablet’s mechanical strength. We need to understand how tablet hardness testing works. This test evaluates how strong a tablet will be when it is mechanically put into production and will be able to withstand the next step in production. A tablet’s strength is associated with its quality characteristics – like how quickly a drug dissolves and how stable it is – which affect how fast a drug will dissolve and how long the patient will be able to maintain the same level of effectiveness from using that drug. Additionally, the relationship between a tablet’s hardness, how well it will be able to maintain its integrity while being coated and release behaviour is vital foreffective therapeutics. Therefore, tablet compression testing is a cornerstone of tablet formulation testing.
What is a Tablet Hardness Tester
A tablet hardness tester Ontario laboratory instrument, is used specifically to measure the what is tablet breaking force of pharmaceutical dosages. A hardness tester is considered to be a very important measurement tool that every pharmaceutical laboratory testing equipment inventory.
 Definition of Tablet Breaking Force
Definition of Tablet Breaking Force
The mechanical force required to fracture a compressed tablet is referred to as tablet breaking force and is expressed in Newtons (N). This value represents how well the tablet can resist chipping, abrasion, and breaking throughout the manufacturing and shipping process.
Importance in Pharmaceutical Quality Control
Hardness testing is not just an assessment; determining tablet mechanical strength testing also verifies tablet durability during transportation and packaging. Hardness testing also ensures that the dosage of the active ingredient remains the same from batch to batch, and confirms the tablet has maintained its structural integrity, which is critical to reliable dissolution performance. Testing for both of these factors is a critical element of pharmaceutical quality control testing.
H2 Standard Hardness Range for Pharmaceutical Tablets
Although the standard hardness range for pharmaceutical tablets can vary significantly, a target hardness range between 40 N and 100 N. This is not definitive; the targeted hardness depends on the characteristics and formulation of the specific formulation, the compression pressure applied during production, and the size/thickness of the tablets. For example, chewable tablets require much lower hardness than those used for post-market controlled release formulations.
Factors Affecting Tablet Hardness
Tablet hardness is dependent on several variables of manufacture. Key factors affecting tablet hardness include the compression pressure applied by the press, the concentration of binders in the formulation, the granule particle size, the amount of lubricant used, and the moisture content of the powder blend before compression.
Units Used in Tablet Hardness Testing
The units used in tablet hardness testing also reflect the evolution of the pharmaceutical industry and the devices that manufacture them.
Newton (N)
The Newton (N) is the SI unit of measurement for force and is utilized as the standard measurement for most modern digital tablet hardness tester equipment, including the THT500, for its precision and universal scientific acceptance.
Kilogram Force (kgf)
Some older testing devices and some regional specification documents reference testing in the unit of measurement referred to as kilogram force or “kgf”. A “kgf” is very close to (9.81 N); however, there is still some slight discrepancy depending upon where you are located.
Kilopond (kp)
Kilopond is a historical unit of measure for pounds and is very similar to kilogram-force. This unit of measure is also referenced in older European tablet hardness testing devices or other active devices.
How a Tablet Hardness Tester Works
It is also important to understand how tablet hardness testing works to effectively perform your duties in the quality control laboratory.
Tablet Placement
Tablet hardness testing is completed by placing a single sample of tablet between the fixed jaw and the movable jaw of the tablet hardness testing machine Ontario.
Force Application
The instrument initiates a controlled mechanical compression, driving the movable jaw against the tablet at a calibrated speed.
Breaking Force Detection
A high precision load cell continuously calculates the force applied. The tablet breaking force tester Ontario records the accurate peak force at the moment the tablet fractures.
Digital Data Recording
The tablet hardness tester immediately displays the result on a digital screen. The data, including hardness, thickness, and diameter, is stored electronically for statistical analysis, batch records, and quality assurance.
Importance of Tablet Hardness Testing in Pharmaceutical Manufacturing
The tablet hardness testing in pharmaceutical manufacturing is crucial for multiple reasons. It conforms to the mechanical stability of tablets, ensuring they can withstand the mechanical stress of packaging operations without capping or laminating. Furthermore, it indicates whether the tablets will survive within the packaging by determining whether the packaging can tolerate breaking under mechanical strain (for example, from transport) while holding the tablets in a blister pack or bottle.
Pharmaceutical Industry in Ontario
The pharmaceutical sector is a major driver of the Ontario life sciences sector. With significant hubs located across Ontario, including the Toronto biotechnology sector and pharmaceutical industry, Mississauga pharma fabrication (manufacturing) cluster, Ottawa biomedical research and development institutions, and Hamilton’s life sciences research ecosystem. The network of contract manufacturers, contract manufacturing companies(CMOs), Ontario biotechnology companies, and Pharmaceutical Research Laboratories Ontario are very widespread and is represented by companies of all sizes; from early-stage startups to large multinationals.
Applications of Tablet Hardness Testing in Ontario
Pharmaceutical Manufacturing
Used on production lines for batch release and in-process control to ensure every batch of tablets meets pre-defined strength specifications.
Research and Development Laboratories
Scientists in R&D use hardness testers for formulation research, excipient compatibility studies, and stability studies to see how hardness changes over time.
Quality Control Laboratories
Independent and in-house pharmaceutical quality control laboratories Ontario use these testers for batch verification and regulatory compliance testing.
Nutraceutical Manufacturing
The growing supplement industry in Ontario uses tablet strength testing machine Ontario units for testing of dietary supplement tablets, ensuring product quality for consumers.
Key Features of the THT500 Tablet Hardness Tester
High-Precision Breaking Force Measurement
The THT500 is an advanced load cell has been engineered to provide extremely accurate and repeatable results. It is considered to be one of the best tablet compression strength tester in Ontario laboratories.
Multi-Parameter Tablet Measurement
Beyond hardness, this laboratory tablet hardness testing device simultaneously an accurate measurement of the hardness of tablets, it also provides simultaneous measurements of tablet diameter and thickness and their overall dimensional profile, allowing for complete evaluation of each sample as a tablet mechanical strength testing device.
Testing Workflow
Utilizing an automatic cycle of testing, the THT500 increases throughput and eliminates operator-to-operator variability, making it the perfect tablet hardness test equipment for busy laboratories.
Digital Data Management
Integrated data storage and connectivity enable seamless transfer of test results from the THT500 to the laboratory information management system (LIMS) to ensure data integrity and traceability.
Technical Specifications of the THT500
The following table provides a summary of the THT500’s technical specifications, highlighting its ability to serve as a top-quality tablet hardness testing device and tablet mechanical strength tester.
| Specification |
Detail |
| Force Range |
0-500 N |
| Measurement Accuracy |
±0.5% |
| Force Resolution |
0.01 N |
| Tablet Diameter Range |
2-40 mm |
| Tablet Thickness Range |
0.1-25 mm |
| Test Speed |
2-200 mm/min (adjustable) |
| Connectivity |
USB, RS232 |
| Dimensions (W x D x H) |
300 x 200 x 120 mm |
| Weight |
8 kg |
Factors That Affect Tablet Hardness During Compression
To achieve consistent tablet hardness, it is crucial to control the production process closely. Many factors affect hardness when the tablets are compressed. The primary compression force creates the earliest compression forces when the powder is density compressed. During the granulation step, how much binder is used plays a critical role in creating cohesion between the granular particles. The distribution of granule sizes will affect how the particles stack together and how easily they compress. While some lubricant is necessary for the ejection of the tablets from a die, excessive use of this material may compromise the tablet bonding ability. The moisture level of a granule also functions as a binder; however, tablets made from granules that have moisture levels outside an acceptable range may encounter issues.
Tablet Hardness Tester vs Other Tablet Testing Equipment
Hardness Tester vs Friability Tester
A hardness tester is one tool used to test a tablet’s mechanical strength; it determines how much force is required to fracture a tablet. A tablet friability testing machine, conversely, measures a tablet’s ability to withstand abrasion. For tablet mechanical strength testing, both tests should be performed to determine the durability of a tablet based on two separate methods.
Hardness Tester vs Dissolution Tester
Whenever a hardness tester is utilized to measure the integrity of a tablet, a dissolution tester should also be used to assess how quickly the active ingredient(s) will be released from the tablet. Both tests provide complementary information; the hardness of a tablet may affect its dissolution performance, therefore both tests should be included in quality control programs and test support for tablet dissolution testing.
Pharmaceutical Testing Equipment Used in Ontario Laboratories
The comprehensive quality assurance in Ontario’s laboratories is based on various types of specialized instrumentation. The most commonly testing equipment found in Ontario laboratories includes tablet hardness testers, friability testers dissolution testers (for performing drug release profiling of tablets), and tablet disintegration testing apparatus. Together with the various types of pharmaceutical tablet testing equipment, these instruments are the central components of the pharmaceutical quality assurance program and play a critical role in ensuring that pharmaceutical products are safe, effective and of high quali
Pharmaceutical Testing Standards
It is essential to comply with the pharmaceutical testing standards required to obtain a legal market approval for a product. Each product must pass a tablet hardness test based on established Pharmacopeia standards before it is distributed. Each pharmacopeia defines the method of performing a tablet hardness test in precise terms: the United States Pharmacopeia <1217> defines instrument performance characteristics, the European Pharmacopeia 2.9.8 defines the procedure for conducting the test, and the Japanese Pharmacopeia defines the data recording method for the test results. The goal of any tablet hardness tester supplier from Ontario is to help their customers build quality products by providing them with the equipment that meets the stringent regulatory and compliance environment.
Common Tablet Manufacturing Problems Detected by Hardness Testing
Using routine hardness testing as a diagnostic tool can identify some common problems occurring during table production. If the hardness test results are inconsistent, then a weakness in the tablet may exist, which could lead to capping or laminated tablets; an over-compressed tablet may create problems with dissolution; or general usability of the tablets suggests that poor powder flow or formulation imbalance may exist. Early detection through strength testing of the tablets being compressed allows the manufacturer to adjust their process before producing a large quantity of tablets, thereby saving both time and materials.
How to Choose a Tablet Hardness Tester
In Ontario, laboratories interested in obtaining a tablet hardness tester should evaluate equipment based on multiple criteria. The equipment must provide accurate and repeatable measurements to ensure consistent, reliable results. Additionally, the level of automation the tablet hardness tester provides must meet the lab’s throughput requirements. Furthermore, it is essential that the tablet hardness tester is compliant with the pharmacopeia. Several advanced data management features will be critical for laboratories conducting regulated work; therefore, the following features would be required within an acceptable hardness test. Audit trails, user access control and throughput must be maintained through the entire testing process.
Frequently Asked Questions
What hardness should a pharmaceutical tablet have?
There exists no single recommended value however, a common target hardness range for tablets is 40-100 Newtons. The optimal mechanical strength of a tablet depends on its formulation, size, and intended use (i.e., immediately released or chewable).
How does a tablet hardness tester work?
A tablet hardness tester determines the hardness of a tablet by placing a tablet between two jaws. One jaw will consistently move downward, generating the force applied to the tablet until the point at which the tablet fails. The peak force produced by the testing machine during the point of failure is the difference in force applied to the tablet when it failed to maintain the integrity of the dosage form and is recorded as the hardness value of the tablet.
What unit measures tablet hardness?
Tablet hardness is measured in Newtons (N), which is the SI unit of measure for force, although the older generation of testing machines may have utilized kilogram-force (kgf) or kilopond (kp) measurement systems. The newer generation of testing machines generally switched to Newtons for the standard of measurement.
Why is tablet hardness testing important?
The importance of tablet hardness testing lies in the SI unit measure for force, although the older generation of testing machines may have utilized kilogram-force (kgf) or kilopond (kp) measurement systems. The newer generation of testing machines generally switched to Newtons for the standard of measurement.
How many tablets are tested for hardness?
How many tablets are tested for hardness depends on the stage of production and pharmacopeial guidelines. Typically, for quality control, a sample of 10 to 20 tablets is tested per batch to get a statistically significant average and variation.
What industries in Ontario use hardness testers?
Pharmaceutical manufacturing companies Ontartio are the primary users, along with Ontario biotechnology companies, contract research organizations, and quality control laboratories. The nutraceutical industry in Ontario also uses these testers for dietary supplement tablets.
Request a Quote for the THT500 Tablet Hardness Tester in Ontario
Enhance your quality control processes with the THT500, the leading tablet hardness tester Ontario laboratories rely on. Contact us today to request product information, schedule a technical consultation, or arrange a product demonstration at your facility. Get detailed pricing and discover how this <span style=”color:blue”>tablet hardness testing instrument Ontario</span> can optimize your tablet strength verification. Secure your tablet hardness testing equipment Ontario quote now