What hardness should a pharmaceutical tablet have?
Most conventional immediate-release tablets within 40–100 Newton, though acceptable ranges vary by formulation. Chewable tablets use lower hardness, while modified-release products may require higher values.
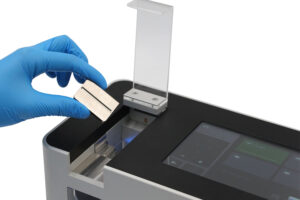
How does a tablet hardness tester work?
The instrument applies a compressive force across a tablet until fracture occurs. A load records the maximum force, which is reported as the breaking force or hardness value.
What unit measures tablet hardness?
Modern pharmaceutical testing uses Newton as the standard unit. Some equipment also displays kilogram force or historical units such as kilopond.
How many tablets are tested for hardness?
5 to 10 tablets are tested per batch or sample lot to obtain significant results, depending on pharmacopeial requirements and internal quality specifications.
Why is tablet hardness testing important?
Tablet hardness testing machine ensures tablets withstand packaging and transport without damage while maintaining appropriate dissolution characteristics. It provides critical quality data for batch consistency and regulatory compliance.
What is the tablet breaking force?
Tablet breaking force is the mechanical force required to fracture a compressed tablet under standardized test conditions, measured in Newtons during diametral compression.
What industries in Florida use hardness testers?
Pharmaceutical manufacturers, contract manufacturing organizations, biotechnology companies, nutraceutical producers, and research laboratories across Miami, Tampa, Orlando, and perform hardness testing.