What hardness should a pharmaceutical tablet have?
There is no universal value, as it depends on the formulation, it varies on the specific formulation. However, many immediate-release compressed tablets fall between 40 to 100 Newtons. The target tablet hardness is determined during the development phase, there’s an adequate amount of strength to resist mechanical stresses during packaging, shipping, handling, and subsequent disintegration without causing any problems for the dissolution and drug release profile.
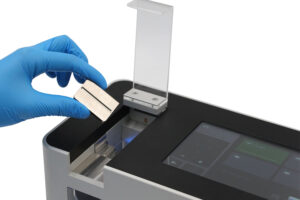
How does a tablet hardness tester work?
To measure the hardness of a tablet, the tablet is placed between the two jaws of an instrument. One jaw exerts a constant speed or force while compressing the tablet until it fractures. A load cell and recorded by the instrument as the peak force required to break the tablet is defined as “hardness” or “breaking/disintegration force”.
What unit measures tablet hardness?
The SI (Standard International) unit for measuring the hardness of a tablet is Newton (N); however some instruments will also provide results in Kilogram-force (kgf) or Kilopond (kp) since the data they generated in the past, were based on these formats and private sector preferences have led to some continued use of these historical units.
Why is tablet hardness testing important?
Tablets must endure the mechanical stresses of packaging, transit, and handling; thus, tablet hardness testing is a critical quality control parameter as well as an assurance of batch-to-batch consistency and that the tablet’s mechanical strength does not negatively impact the dissolution and drug release profiles.
How many tablets are tested for hardness?
Depending on the specific provisions of each pharmacopeia or internal standards, the number of tablets to be tested when sampling for batch release will typically range from 5 to 20 tablets per unit, though larger sample sizes may be appropriate during process validation or troubleshooting to ensure statistical significance.
What industries in California use hardness testers?
The pharmaceutical industry mainly utilizes these instruments and includes industry leaders in the biotech hub in San Diego, as well as one-third of all biotech companies based in the Bay Area. Other common users of these instruments include Contract Manufacturing Organizations (CMOs), biomedical research laboratories, and nutraceuticals (dietary supplement products).

 Definition of Tablet Breaking Force:
Definition of Tablet Breaking Force: